Impact Story • Success Story
Helping Babies Suffering from Opioid Withdrawal at Birth
By Lisa C. Welch(1), et al.
All Authors and Affiliations
Affiliations:
1. Tufts University Boston
2. Tufts Medical Center
Posted April 21, 2026
Featured CTSA Institutions:
Tufts Medical Center
Abstract
From the 1990s to today, a growing number of babies are exposed to an opioid before birth and have withdrawal soon after, a condition called neonatal abstinence syndrome. They have painful symptoms and may need to be in the hospital for weeks. Before 2017, there was no research on which medicines best calm these babies. When Dr. Jonathan Davis and his team set out to change that, Tufts Clinical and Translational Science Institute (CTSI) helped by designing a clinical trial and analyzing data from many hospitals. Results showed that a different medicine (methadone)--not the one used most often (morphine)--led to fewer days of treatment and shorter hospital stays. This became part of the American Academy of Pediatrics treatment guidelines. Tufts CTSI also assisted Dr. Davis as he worked to change research rules and laws to solve barriers to doing the study. This led Dr. Davis to take part in developing best practices for including newborns in health research that guide today’s studies.
2-3
Days shortened treatment and hospital stay
2
New guidelines informed
2
New laws contributed to
Impact Story

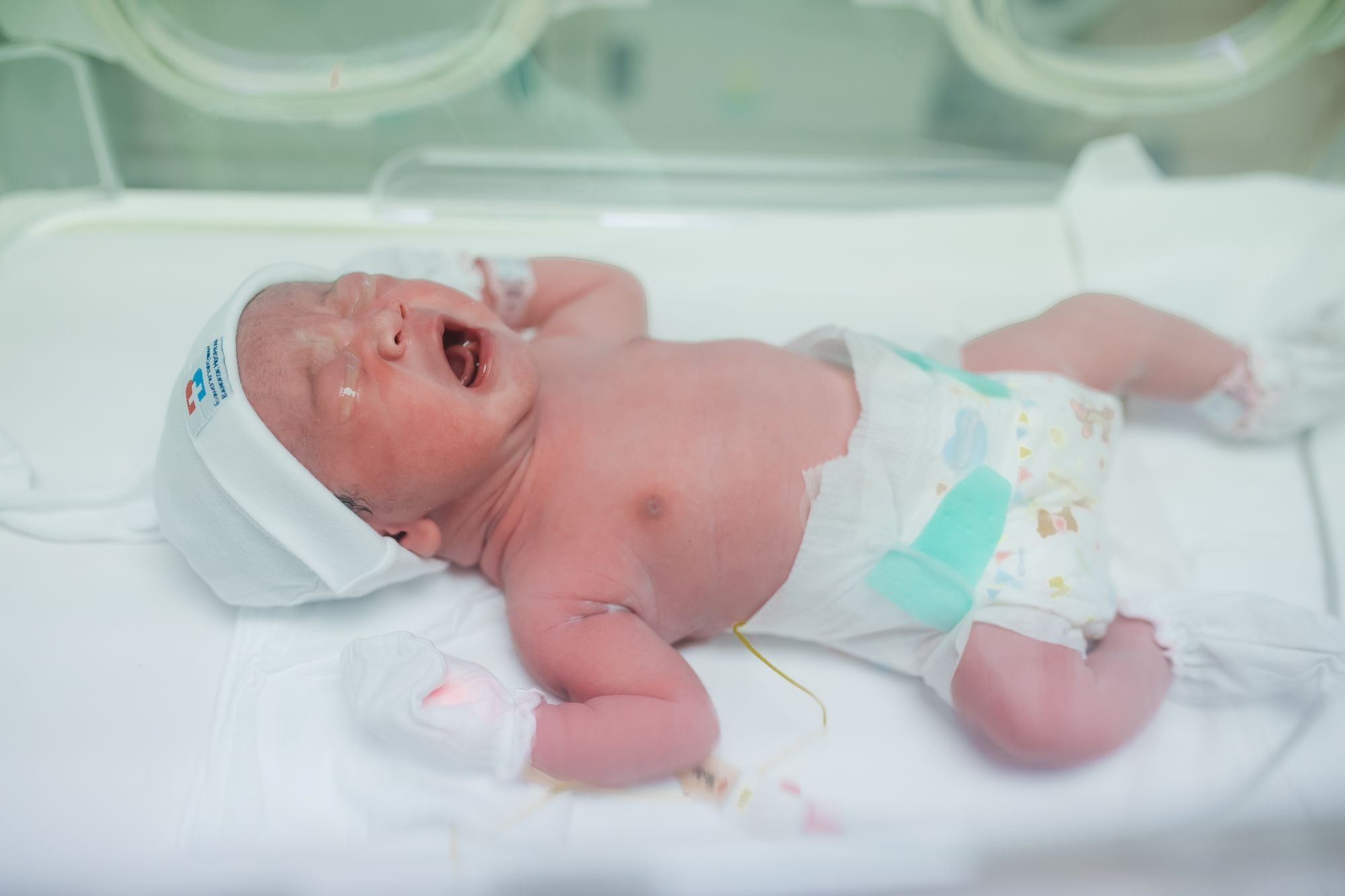
Starting in the late 1990s and continuing today, a growing number of babies develop withdrawal soon after birth, a condition called neonatal abstinence syndrome (NAS). They were exposed to an addictive drug before birth (usually an opioid like heroin, fentanyl, methadone, or even prescription painkillers) and begin going through withdrawal after delivery.
These babies have many painful symptoms, cry often, and may need to stay in the hospital for weeks. A recent estimate showed that more than six out of every 1,000 babies have significant withdrawal soon after birth. This means that more than one in every 200 babies in the U.S. is born with withdrawal that’s about one every 15 to 20 minutes.
Doctors and nurses do all they can to help these babies, and health research has played a big role in finding the best medicines and treatments to use. Before 2017, there was no research showing what medicines are best for babies born with withdrawal. Doctors were using medicines created for adults (most often morphine) and estimating the right doses for their youngest patients.
A team led by Jonathan Davis, M.D. at Tufts Medical Center set out to change this. In 2012, this team led the first large study with newborns to compare the most common drug being used to help calm babies with symptoms of withdrawal (morphine) to a longer-acting medicine (methadone). At the time, the team faced many challenges with this study. Doing research with a controlled drug led to legal hurdles. The social stigma around drug use and the side effects of many treatments for opioid use made it hard for mothers to join the study. Plus, doing research with newborns was not common because parents and government agencies were worried about risks to fragile babies.
Tufts Clinical and Translational Science Institute (CTSI) helped make this research possible. Tufts CTSI provided support to carefully design the study and receive money from the National Institutes of Health to run it at many hospitals across the country. Tufts CTSI statisticians also analyzed a large amount of information from those hospitals. Finally, Tufts CTSI assisted Dr. Davis as he began speaking with lawmakers and government agencies about changing laws and research rules so that the team could complete the study.
Study results were surprising. The longer-acting medicine (methadone), not the accepted treatment (morphine), led to fewer days of treatment and fewer days in the hospital. Around the same time, another research team showed that a similar longer-acting medicine (buprenorphine) also led to fewer days of treatment and shorter hospital stays compared to morphine.
"This work has been ‘all about the relationships’… [If] you want to do great science, you must work well [together].”
- Dr. Jonathan Davis
These results helped improve how doctors treat babies born with withdrawal. The American Academy of Pediatrics published a new guideline recommending longer-acting medicines like methadone instead of morphine to shorten treatment time and hospital stays. Shorter hospital stays get babies home to their families sooner and also can lower health care costs.
“Connecting clinical care, research, and advocacy is key to making an impact. The advocacy part reinforces that you need to do more research, which in turn supports the most effective clinical care.”
- Dr. Jonathan Davis
Just as important, the research led to many more studies about what treatments are best for newborns with opioid withdrawal. Dr. Davis worked with government agencies and policymakers to pass new laws to help families and make sure treatments for newborns are safe and effective. These include the FDA Safety and Innovation Act of 2012 that required the Food and Drug Administration to hire more experts in newborn medicine and the Protecting our Infants Act of 2015 to help families of babies with withdrawal after birth. He also worked with partners in the U.S. and other countries to develop best practices for doing research with newborns.
What began as a question by doctors at one hospital about how to best help babies with opioid withdrawal after birth grew into a national effort to improve care for some of the youngest and most vulnerable patients that continues today.



